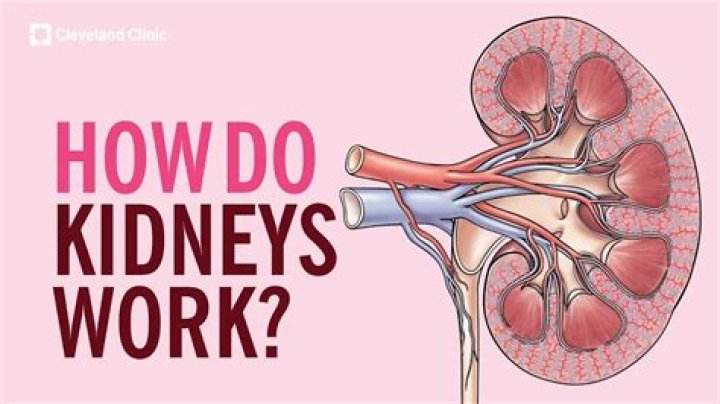
Iodine has seven valence electrons. Valence electrons of an atom are located in the outermost shell of the atom and participate in bonding.
How many electrons are there in valence shell of iodine?
Iodine has 7 valence electrons in the valence shell which means that there are 7 electrons in its outer shell. A neutral iodine atom will have a total of 53 electrons around its nucleus.
What is the outermost shell of iodine?
Iodine has an electron configuration of [Kr]4d105s25p5, with the seven electrons in the fifth and outermost shell being its valence electrons.
How many electrons are there in iodine?
I is iodine. Iodine is element number 53. Iodine has 53 protons and 53 electrons (because it has no charge the number of protons equals the number of electrons).How many valence electrons does gallium?
Gallium has the following electron configuration. The 4s and 4p electrons can be lost in a chemical reaction, but not the electrons in the filled 3d subshell. Gallium therefore has three valence electrons.
How do you find the electrons?
To calculate the numbers of subatomic particles in an atom, use its atomic number and mass number: number of protons = atomic number. number of electrons = atomic number.
How do you figure out valence electrons?
For neutral atoms, the number of valence electrons is equal to the atom’s main group number. The main group number for an element can be found from its column on the periodic table. For example, carbon is in group 4 and has 4 valence electrons. Oxygen is in group 6 and has 6 valence electrons.
What is the longhand electron configuration for iodine?
Using our example, iodine, again, we see on the periodic table that its atomic number is 53 (meaning it contains 53 electrons in its neutral state). Its complete electron configuration is 1s22s22p63s23p64s23d104p65s24d105p5. If you count up all of these electrons, you will see that it adds up to 53 electrons.How many valence electrons are in Argon?
-So, the number of electrons in the Argon atom is 18 because it is present in the neutral state. Hence, from the above given configuration it is clear that the number of valence electrons in the Argon atom is eight.
How many valence electrons does iodine and chlorine have?Iodine has 7 electrons in the outer shell and Chlorine has 7 electrons in its outermost shell so will gain 1 more to complete the octet.
Article first time published onHow many electrons are there in CA?
Ca has 20 protons, so neutral it would have 20 electrons, but according to the charge, 2 electrons have been lost.
What is iodine on the periodic table?
iodine (I), chemical element, a member of the halogen elements, or Group 17 (Group VIIa) of the periodic table.
How many outer shell electrons does gallium have?
Gallium atoms have 31 electrons and 31 protons with 3 valence electrons in the outer shell. Under standard conditions gallium is a soft metal with a silvery color.
How do you find the number of electrons in the outer shell?
So we can use the column or group of an element to determine the number of electrons in its outermost shell, sometimes referred to as valence electrons. Elements from the fourth column of the periodic table, like carbon, have four electrons in their outermost electron shell or four valence electrons.
How many electrons are in each shell?
Energy shellMaximum number of electronsFirst2Second8Third8
How many electrons are gained or lost in iodine?
Iodine is more likely to gain one electron. It will become I− ion.
What is the electron number?
Finding the Number of Electrons The number of electrons in an atom is equal to the atomic number of an element, for neutrally charged species. This means the number of electrons and the number of protons in an element are equal. Therefore, the number of electrons in oxygen is 8.
How do you find the neutron number?
To find the number of neutrons, subtract the number of protons from the mass number. number of neutrons=40−19=21.
How many valence electrons are in Sulfur?
The superscripts associated with these orbitals total to 6. Therefore, sulfur has 6 valence electrons.
What is Argon valence shell?
Explanation: Argon is a noble gas with an electron configuration of [Ne]3s23p6 . So, it has 3 electron shells, and that will be its valence shell. In the third shell, it holds a total of 2+6=8 electrons, so it has 8 valence electrons.
Is Argons valence shell full?
Although argon does not technically have a full outer shell, since the 3n shell can hold up to eighteen electrons, it is stable like neon and helium because it has eight electrons in the 3n shell and thus satisfies the octet rule.
How many electrons are in the outer shell of argon?
The complete octet (eight electrons) in the outer atomic shell makes argon stable and resistant to bonding with other elements.
Does iodine have 5 valence?
Iodine has seven valence electrons. Valence electrons of an atom are located in the outermost shell of the atom and participate in bonding. There are 7 valence shell electrons and 46 inner shell electrons in the ground state of iodine.
How many valence electrons does an iodine atom have reason it?
Explanation: Iodine is in the group 17 elements, known as halogens, and all of them have 7 valence electrons. A valence electron is capable of bonding with valence electrons of another atom to complete the octet of electrons each atom needs in its outer shell.
How many valence electrons are in Ca2 +?
Since we are talking about the 2+ cation, it already lost two electrons. We can tell because each electron brings a 1− charge, and so losing a 1− charge is like gaining a 1+ charge. Also, since neutral Ca is on the second column/group, it originally had 2 electrons. 2−2=0 , so Ca2+ has no valence electrons .
How many electrons are containing in Ca2+?
Ca2+ represents an ion with 20 protons and 18 electrons. A calcium atom has 20 protons and 20 electrons. The 2+ charge next to the symbol indicates a loss of two electrons: 20-2=18. When atoms form ions, they lose or gain electrons.
How many valence electron does bromine have?
Valence shell configuration of bromine is 4s24p5. Therefore Bromine has 7 valance electrons.
Is Iodine an ion?
NamesChemical formulaI −Molar mass126.90447 g·mol−1Conjugate acidHydrogen iodideThermochemistry
Is Iodine a base?
Iodine is neither an acid NOR a base…..
How do we find atomic mass?
For any given isotope, the sum of the numbers of protons and neutrons in the nucleus is called the mass number. This is because each proton and each neutron weigh one atomic mass unit (amu). By adding together the number of protons and neutrons and multiplying by 1 amu, you can calculate the mass of the atom.