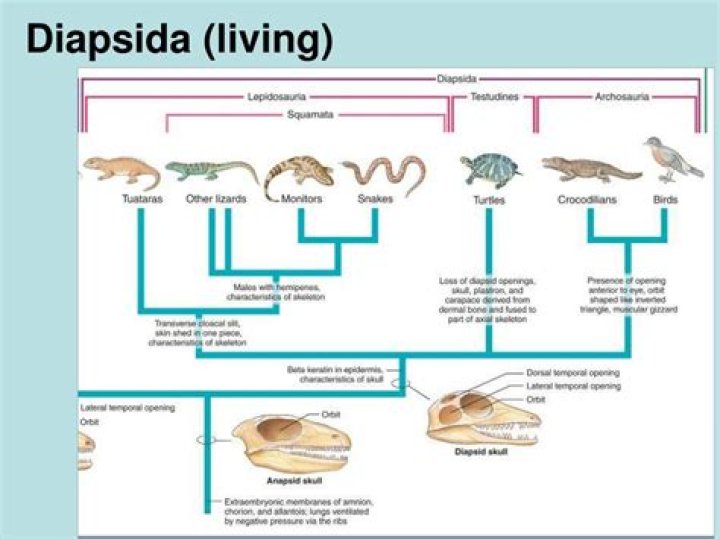
Ammonia (NH 3) is a base even though it does not contain OH − ions in its formula.
Can NH act as a base?
15.13 Amines as Bases Recall that ammonia (NH 3) acts as a base because the nitrogen atom has a lone pair of electrons that can accept a proton. … Nearly all amines, including those that are not very soluble in water, will react with strong acids to form salts soluble in water.
Is NH2 1 an acid or base?
Ammonia is actually itself a weak base, so its conjugate base NH2- is an incredibly strong base so it can get an extra proton to regenerate NH3 which is much more stable. As a result of the relative basicity of NH2- and Cl-, we can determine that acid chlorides are much more reactive than amides.
Is NH3 always a base?
NH3 will always be a base. In fact it is a weak base.Is NH an amino group?
Reaction nameReaction productCommentcarbylamine reactionIsonitrilePrimary amines only
Is RbOH strong base?
Strong bases are capable of deprotonating weak acids; very strong bases can deprotonate very weakly acidic C–H groups in the absence of water. RbOH is Rubidium hydroxide, it is a strong base.
Can NH2 act as an acid?
NH2 is acidic in nature as NH2 is a group is it is a functional group which is generally present in the form of NH2 plus that is cationic form NH2 Plus shows the more tendency to accept electrons which makes it acidic in nature as the acid the tendency to accept electron.
Is NH a strong base?
Strong baseWeak baseThey ionize completely.They do not completely ionize.Can ammonia act as an acid?
Ammonia is normally a base, but in some reactions it can act like an acid. Ammonia acts as a base. It accepts a proton to form ammonium. Ammonia also acts as an acid.
Is NH more acidic than Oh?#3 Importance – all things being equal, an OH acid is more acidic than an NH acid. This principle can be very useful if used properly.
Article first time published onWhat is the conjugate acid of NH2?
Hence, for NH2- the conjugate acid would be NH3 (the positive charge of H+ and the negative charge of NH2- cancel each other out).
What functional group is NH?
NAMEGROUPBIOLOGICAL EXAMPLEamine:-NH2amino acid (e.g., glycine)detail:(-NH3+)ketone:-CO-metabolic intermediate (e.g., pyruvic acid)detail:O || -C-
How many hydrogens are attached to the nitrogen in a tertiary amine?
-Primary amine (1°) – nitrogen atom is bonded to one hydrocarbon group and two hydrogen atoms. -Secondary amine (2°) – nitrogen atom is bonded to two hydrocarbon groups and one hydrogen atoms. -Tertiary amine (3°) – nitrogen atom is bonded to three hydrocarbon groups and no hydrogen atoms.
Are amino acids amides?
The amide group is called a peptide bond when it is part of the main chain of a protein, and an isopeptide bond when it occurs in a side chain, such as in the amino acids asparagine and glutamine. … Amides include many other important biological compounds, as well as many drugs like paracetamol, penicillin and LSD.
Can H2O act as a base?
Answers. Under the right conditions, H2O can donate a proton, making it a Brønsted-Lowry acid. Under the right conditions, H2O can accept a proton, making it a Brønsted-Lowry base.
What is the conjugate base of NH3?
NH2− is the conjugate base of NH3.
Does F have a conjugate base?
The substance that is produced after an acid has donated its proton is called the conjugate base while the substance formed when a base accepts a proton is called the conjugate acid. … Since the product H3O+ can donate a proton back to F– it is labeled the conjugate acid, while the F– is the conjugate base.
Is FA Lewis an acid or base?
A Lewis base, then, is any species that donates a pair of electrons to a Lewis acid to form a Lewis adduct. For example, OH− and NH3 are Lewis bases, because they can donate a lone pair of electrons. You can now imagine why F is a Lewis base.
Is NH2 a weak acid?
The conj. base of NH3 acting as an acid is NH2^-. This makes NH2^– a strong base.
Why is sncl4 a Lewis acid?
SnCl4 is a lewis acid. This is because chlorine being the more electronegative atom, pulls the shared pair of electrons towards itself, making tin electron deficient. As a result, Sn can accept electron pairs from molecules like NH3, thereby forming a coordinate bond.
Is LIBR a strong acid?
Salt formed from:Salt SolutionWeak acid + Strong baseBasic
Is RbOH an acid base or salt?
AcidsBasesHNO3RbOHH2SO4CsOHHClO3Mg(OH)2HClO4Ca(OH)2
Is RbOH an acid in water?
HBrO is a weak acid, so BrO– is a weak base, but not very weak.
Is Vinegar a base?
Substances over 7 are categorized as basic, with 7 being the neutral point. Water has a pH level of 7. Substances with pH levels under 7 are categorized as acidic. Vinegar is acidic.
Is milk an acid or base?
Cow’s milk Milk — pasteurized, canned, or dry — is an acid-forming food. Its pH level is below neutral at about 6.7 to 6.9. This is because it contains lactic acid. Remember, though, that the exact pH level is less important than whether it’s acid-forming or alkaline-forming.
Is bleach a base?
Chlorine bleach is a base and is especially good at removing stains and dyes from clothes as well as disinfecting.
Is HOAc an acid?
Illustrated Glossary of Organic Chemistry – Acetic acid; Ethanoic acid; HOAc; AcOH; vinegar. Acetic acid (Ethanoic acid; HOAc; AcOH): A nonpolar (ε = 6.2) protic solvent and moderately weak acid (pKa = 4.78) of molecular formula C2H4O2. IUPAC name ethanoic acid.
Is HCN a strong acid?
AcidConjugate BaseHCN (hydrocyanic acid) (weakest)CN− (cyanide ion) (strongest)
Is Hcooh a strong acid?
Formic acid (HCOOH) is a weak acid. We know it is an acid because it contains the carboxyl (COOH) functional group, which makes it a carboxylic acid….
Is NH a stronger base than O?
The conjugate acid of (a) is H3O+ which has a pKa of -2 and the conjugate acid of (b) is NH4+ which has a pKa of 9. … The conjugate acid of (b) is a carboxylic acid which has a pKa of 5. Therefore, (a) is the stronger base because it has a weaker conjugate acid. ANSWER: The lone pairs on oxygen (a) are the most basic.
How basic is NH2?
NH2- Acid or Base NH2- is a strong base because it is unstable with its negative charge in a solution so that it wants to take the edge off with a negative charge by accepting a proton and acting as a base.