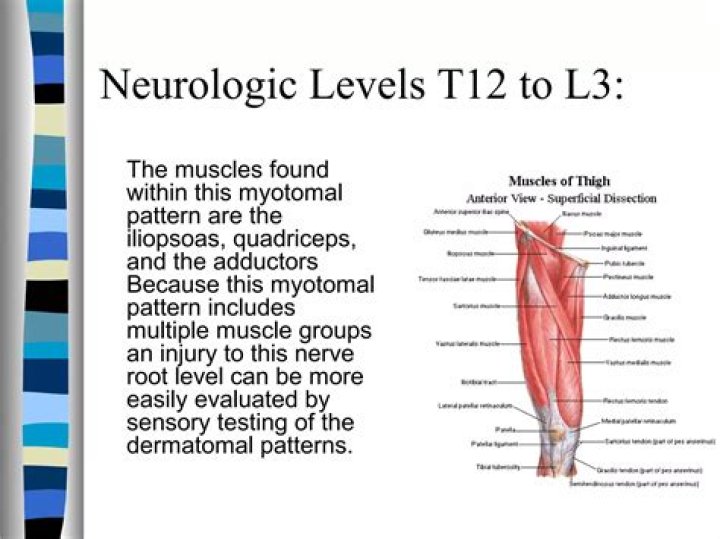
IdentifiersSolubility in water535 g/L (0 °C) 678 g/L (25 °C) 1020 g/L (100 °C)Solubilityvery slightly soluble in diethyl etherSolubility in glycerol217 g/L
Is potassium bromide soluble in water or oil?
IdentifiersSolubility in water535 g/L (0 °C) 678 g/L (25 °C) 1020 g/L (100 °C)Solubilityvery slightly soluble in diethyl etherSolubility in glycerol217 g/L
Is potassium bromide soluble in acetone?
Conclusions. The solubility of KBr in water−acetone solvent was obtained experimentally. The solubility of KBr in water and acetone solvent increases with an increase in temperature and with a decrease in the concentration of acetone.
How do you dissolve potassium bromide?
Potassium bromide is dissolved in double distilled water to form a solution of 200 or 250 mg/ml.What is potassium iodide soluble in?
Potassium iodide is highly soluble in water so SSKI is a concentrated source of KI. At 20 degrees Celsius the solubility of KI is 140-148 grams per 100 grams of water.
Is nano3 soluble?
Sodium nitrate is a white deliquescent solid very soluble in water.
Is potassium bromide soluble in water or heptane?
Potassium bromide is more soluble in water.
What type of bonding is present in potassium bromide?
Potassium bromide has ionic bonding between its two elements potassium and bromine. Bromine has -1 charge, and potassium has a +1 charge.Is potassium bromide soluble in ethanol?
Potassium bromide is slightly soluble in ethanol, glycerol and alcohol.
What is the correct name of KCL?ChEBI Namepotassium chlorideChEBI IDCHEBI:32588DefinitionA metal chloride salt with a K(+) counterion.
Article first time published onIs nh4oh soluble?
Ammonium hydroxide is an inorganic herbicide, fungicide and microbiocide.It is non-volatile and highly soluble in water.
Is cucl2 soluble?
Copper(I) chloride, commonly called cuprous chloride, is the lower chloride of copper, with the formula CuCl. The substance is a white solid sparingly soluble in water, but very soluble in concentrated hydrochloric acid.
Is potassium iodide soluble in hexane?
Potassium iodide, an ionic compound, dissolves easily in water but does not dissolve in chloroform and hexane.
Is potassium iodide soluble in ethanol?
Potassium Iodide is soluble in ethanol at about 1.8g/100mL at room temp.; follow the link to the reference.
Is heptane water soluble?
Heptane (CAS 142-82-5) is principally derived from crude oil and classified as a straight-chain neutral aliphatic hydrocarbon. It is a colorless, volatile petroleum distillate that is relatively insoluble in water and is present as a major component of gasoline.
Why is KBr not soluble in hexane?
The given compounds, hexane, benzene, cyclohexane, and carbon tetrachloride, are non-polar compounds; therefore, they do not have a driving force for ionic attraction. Hence, they are unable to dissolve KBr.
How is MgCl2 soluble?
MgCl2 is an ionic and thus polar compound. So, it dissolves in polar water but insoluble in non- polar acetone. MgCl2 is an ionic compound. Thus it can easily dissolve in polar solvents like water (like dissolves like).
Why is NaNO3 soluble?
A polyatomic ion does not break up into separate atoms in solution. Thus, a solution of sodium nitrate, NaNO3, contains sodium ions and nitrate ions; no nitrogen ions or oxygen ions are present. … This dissolution of ionic compounds takes place because of interactions like that between ammonia and water.
What is NaNO3?
Sodium nitrate (NaNO3)
Is NaNO3 aqueous or solid?
CHEMICAL NAMEPHYSICAL STATEFORMULA / SYMBOLsodium nitratesolidNaNO3sulfursolidSsulfur dioxidegasSO2sulfuric acidliquidH2SO4
Is HBr insoluble?
HBr is very soluble in water, with solutions of HBr in water forming hydrobromic acid which dissociates to ions (H+and Br-).
What is HBr water?
Hydrobromic acid is a strong acid formed by dissolving the diatomic molecule hydrogen bromide (HBr) in water. “Constant boiling” hydrobromic acid is an aqueous solution that distills at 124.3 °C and contains 47.6% HBr by mass, which is 8.77 mol/L.
Is potassium bromate banned in Canada?
Potassium bromate is also illegal in the European Union, Canada, Brazil and elsewhere because it causes cancer in rats and mice. In the United States, however, it has remained legal since it was first patented for use in baking bread, in 1914.
What are bromide salts?
Sodium bromide is a white, crystal-like, compound made of sodium and bromide (a charged bromine atom). It’s appearance closely resembles normal table salt. It is commonly used for the bromide that it contains, which means that this compound has a wide variety of uses.
Is caco3 soluble in water?
Calcium carbonate appears as white, odorless powder or colorless crystals. Practically insoluble in water. Occurs extensive in rocks world-wide.
What type of bond is MgCl2?
MgCl2 is an ionic compound. Magnesium is a metal with a positive charge of +2.
Is potassium bromide a covalent bond?
The bond between K and Br in KBr is considered ionic. … Covalent bonds occur when electrons are shared between atoms rather than transferred.
Is Ca Oh 2 ionic or covalent?
Calcium hydroxide consists of an ionic bond between a calcium ion, Ca2+ and a hydroxide ion, OH− . The hydroxide ion itself has a single covalent bond between the oxygen and the hydrogen atoms, and so it is a covalent compound ion.
What is in potassium permanganate?
What is potassium permanganate? Potassium permanganate is a common chemical compound that combines manganese oxide ore with potassium hydroxide. It was first developed as a disinfectant in 1857. Since then, it’s been widely used to treat a variety of skin conditions, including fungal infections.
What is FeCl2 in chemistry?
Iron(II) chloride, also known as ferrous chloride, is the chemical compound of formula FeCl2. It is a paramagnetic solid with a high melting point.
Why is potassium chloride soluble in water?
2: As potassium chloride (KCl) dissolves in water, the ions are hydrated. … Ion-dipole forces attract the positive (hydrogen) end of the polar water molecules to the negative chloride ions at the surface of the solid, and they attract the negative (oxygen) ends to the positive potassium ions.