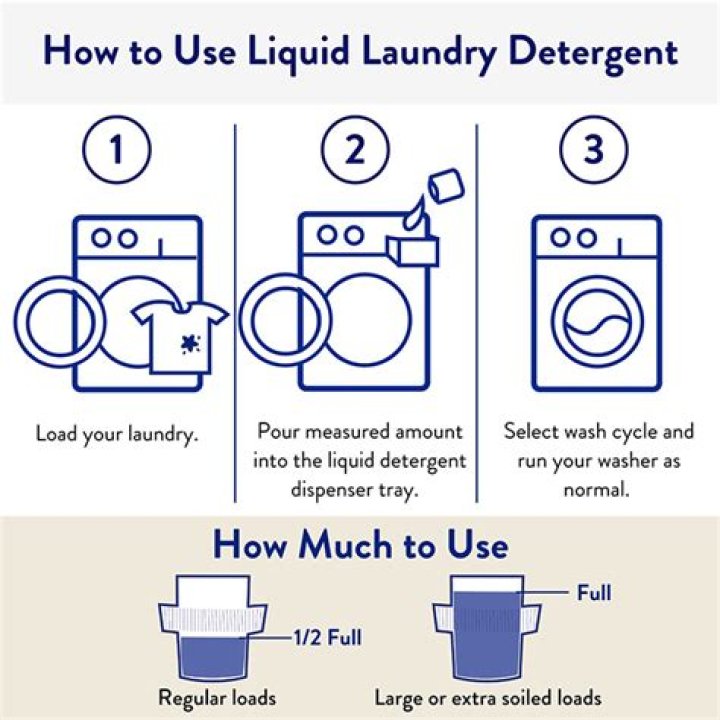
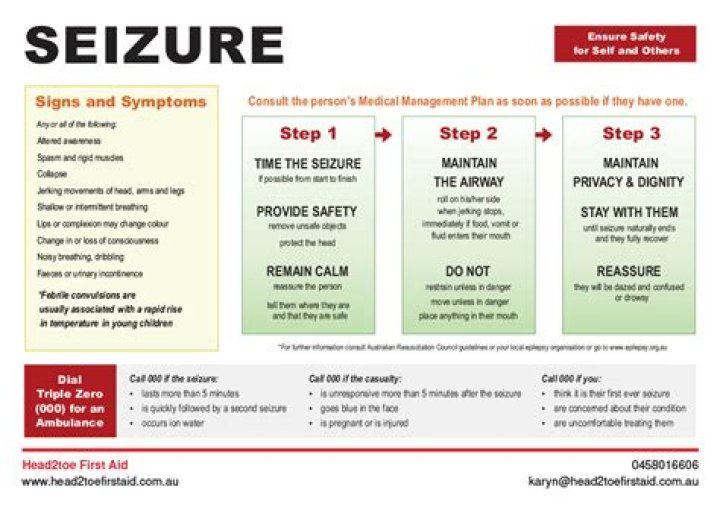
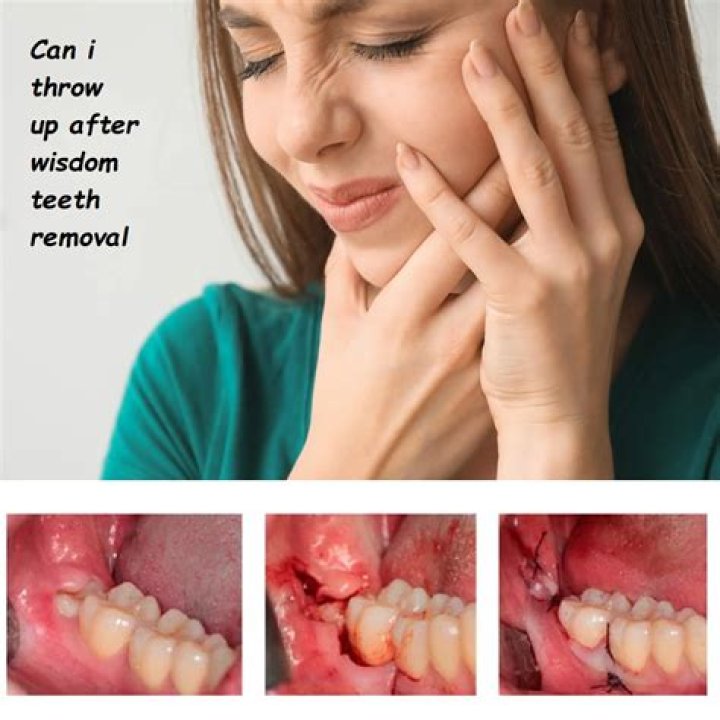
Which Formulas Represent One Ionic Compound And One Molecular Compound
Which formula represents an ionic mixture and a molecular mixture? 3
(1) N2 and SO2
(2) Cl2 and H2S
(3) BaCl2 and N2O4
(4) NaOH and BaSO4
Please explain the reason
Ionic compounds are compounds formed by ionic bonds. All of these compounds are molecular compounds in that they are all made up of molecules. Synchronous and ionic bonds are molecular bonds. If you know which one will form the ionic bond with the other, you will be fine.
BaCl2 and N2O4