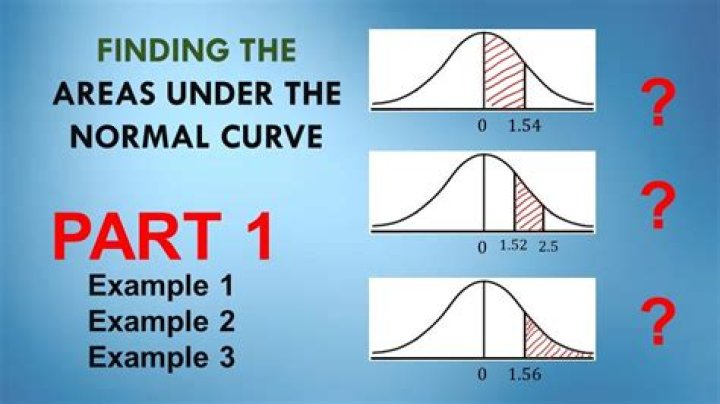
PCl3 is not trigonal planar. It is trigonal pyramidal in shape. This is due to PCl3 being sp3 hybridized.
Why does PCl3 have pyramidal shape?
Looking at the PCl3 molecular geometry it is trigonal pyramidal with a bond angle of approx. 103o. The is mainly due to the disproportionate influence or greater repulsion of the phosphorus lone pair which makes it deviate from the ideal angle of 109o.
What makes a trigonal pyramidal?
Central atom will at the apex and three other atoms or ligands will be at one base, where they are in the three corners of a triangle. There is one lone pair of electrons in the central atom. … So when the position of the lone pair is neglected, the remaining bonds make the trigonal pyramidal geometry.
Why is ph3 trigonal pyramidal?
Explanation: There are 5+3 valence electrons for which to account; and this gives 4 electron pairs arranged the central phosphorus atom. These assume a tetrahedral geometry, however, one of the arms of the tetrahedron is a lone pair, and geometry descends to trigonal pyramidal with respect to phosphorus.What is the molecular geometry for PCl3?
Central atom:P3 x Cl contibute:3 e−Total VSE:8Total VSEP:4Geometry:Trigonal pyramidal (based on tetrahedral)
Is PH3 trigonal planar or pyramidal?
Answer: The PH3 P H 3 molecule is (d) Trigonal bipyramidal.
Why is PCl3 not trigonal planar?
PCl3 is not trigonal planar. It is trigonal pyramidal in shape. This is due to PCl3 being sp3 hybridized.
Why does a trigonal pyramidal molecule have a different shape than a trigonal planar molecule?
This is because the lone pair electrons repel the bonded electrons, giving it a trigonal pyramidal shape. On the other hand, when there’s no lone pair, they form the trigonal planar shape because they aren’t being repelled by that lone pair.Why is the bond angle of PH3 93?
PH3 has a bond angle closer to 90° because of poor overlap between the s and p orbitals, so there is little hybridization of orbitals. This causes the lone pair of PH3 to take nearly all of the s orbital with little p mixed in, and the remaining orbitals (for P-H bonds) are nearly pure p orbitals.
Which one has a trigonal pyramidal structure?The ammonia molecule has a trigonal pyramidal shape with the three hydrogen atoms and an unshared pair of electrons attached to the nitrogen atom. It is a polar molecule and is highly associated because of strong intermolecular hydrogen bonding.
Article first time published onWhy is PCl3 polar?
The PCl3 molecule is polar in nature due to its asymmetric form and difference in electronegativity. The formal charge on phosphorus of PCl3 molecule is zero. The dipole moment of PCl3 molecule is 0.97 D.
Is PCl3 ionic or covalent?
Phosphorus trichloride (PCl3 ) is covalent molecule.
What is trigonal planar in chemistry?
In chemistry, trigonal planar is a molecular geometry model with one atom at the center and three atoms at the corners of an equilateral triangle, called peripheral atoms, all in one plane. In an ideal trigonal planar species, all three ligands are identical and all bond angles are 120°.
Why is PH4+ tetrahedral?
P in both PH3 and PH4+ is hybridized. Due to the absence of lone pair-bond pair repulsion and presence of four identical bond pair-bond pair interactions, PH4+ assumes tetrahedral geometry with a bond angle to 109° 28′.
Why PH4 has more bond angle than PH3?
In PH3 P is sp3 hybridized. Three orbitals are involved in bonding with three hydrogenatoms and the fourth one contains a lone pair. … Due to the absence of lone pair in PH+4 there is no lone pair-bond pair repulsion. Hence the bond angle PH+4 in is higher than the bond angle in PH3.
Does PH4+ Have coordinate bond?
But in case of PH4+ the lone pair of phosphorus is donated to the H+ (hydronium ion) for making a coordinate bond. Thus there is not any lone pair on phosphorus atom. As the bond pair-bond pair repulsions are quite lesser than lone pair-bond pair so it gets proper tetrahedral structure. … The structure is as shown below.
Why is the bond angle of PH3 less than PCl3?
bond angle is increased. Hydrogen atoms are smaller than chlorine, so the PH3 cone will be smaller than that for PCl3. Therefore, the H-P-H bond angle will be smaller than the Cl-P-Cl bond angle.
What are the bond angles in the PH3 molecule likely to be?
Phosphine is the 2nd row analogue of ammonia. There are 4 regions of electron density around the phosphorus atom, 1 of which is a lone pair. ∠H−P−H bond angles should be < 109.5∘ , approx. 105−7∘ .
What is the bond angle of AsH3?
NH3 has bond angle of 107°and PH3 and AsH3 possesses respectively bond angle of 93° and 91°.
What causes a trigonal pyramidal molecule to not be flat?
The nonbonding pair of electrons pushes away from the bonding pairs producing a trigonal pyramidal shape. If the central atom with no lone pair is bonded to three other atoms the molecule will have a trigonal planar shape.
Is pyramidal the same as trigonal pyramidal?
Trigonal Bipyramidal vs Trigonal Pyramidal Both are pyramidal in shape, and both can have similar central atoms, The differences pile up, however. The molecules that form the two shapes have different steric numbers, and they utilize different hybridizations.
Why is Hydronium trigonal pyramidal?
Re: H3O+ The Lewis structure has 8 electrons, and when drawing the Lewis structure there are three bonds to the oxygen, with one lone pair. So there are 4 electron domains, so the molecular geometry is tetrahedral. But one electron domain is a lone pair, so the actual shape is trigonal pyramidal.
What causes trigonal planar?
A trigonal planar molecule has a central atom bonded to three surrounding atoms, with no lone electron pairs. Therefore, its steric number (the total number of bonds and lone electron pairs) is three. The bonds form angles of 120 degrees.
Why is trigonal planar?
A compound with trigonal planar molecular geometry has a central atom bonded to three other atoms or groups. As it has no lone pairs of electron pairs, the three groups to which it is bonded are arranged like a triangle around the central atom, with the bond angles measuring 120 degrees.
Why do trigonal pyramidal molecules have a smaller bond angle than tetrahedral molecules?
(b) The trigonal pyramidal molecular structure is determined from the electron-pair geometry. (c) The actual bond angles deviate slightly from the idealized angles because the lone pair takes up a larger region of space than do the single bonds, causing the HNH angle to be slightly smaller than 109.5°.
Does trigonal have pyramidal geometry?
Trigonal pyramidal molecular geometryExamplesNH3Point groupC3vCoordination number3Bond angle(s)90°<θ<109.5°
How is the trigonal pyramidal molecular geometry related to the tetrahedral electron geometry?
A tetrahedral is a kind of pyramidal structure that has four “equal” triangular sides or faces (four identical atoms). A trigonal pyramid, on the other hand, has one lone atom and three identical atoms at its corners.
What is reason amines become pyramidal geometry?
Amines typically have three bonds and one pair of lone pair electrons. This makes the nitrogen sp3 hybridized, trigonal pyramidal, with a bond angle of roughly 109.5o.
Is pcl3 a molecular compound?
Phosphorus trichloride is a chemical compound. It is a molecular compound made up of phosphorus covalently bonded to three chlorine atoms. The phosphorus is considered to be in a 3+ oxidation state and each chlorine is considered to be in a 1- oxidation state. The chemical formula is PCl3.
Does pcl3 obey octet rule?
In PCl3, the octet for both phosphorus and Chlorine atoms is complete. Therefore, it follows the octet rule.